Laboratory: Lipid vesicles
Lipid vesicles can easily be prepared from a number of different lipids that are present in biological membranes or a number of different non-natural, fully synthetic surfactants. Membrane proteins can be reconstituted in liposomes, allowing the investigation of membrane embedded proteins in a biomimicking environment, outside the living cell. The size of the vesicles prepared can be controlled to some extent, varying between less than 50 nm and more than 10 μm, allowing studies of certain fundamental aspects of organelles and cells as a whole. There are many methods which allows preparing relatively defined vesicular suspensions with acceptable chemical and colloidal stability (size, size distribution, lamellarity) from the simpler consisting of a lipid-film hydration, trough the extrusion to prepare large unilamellar vesicles, detergent dialysis method and to electroformation technique or emulsion inversion method to prepare giant vesicles visible under optical microscopy.
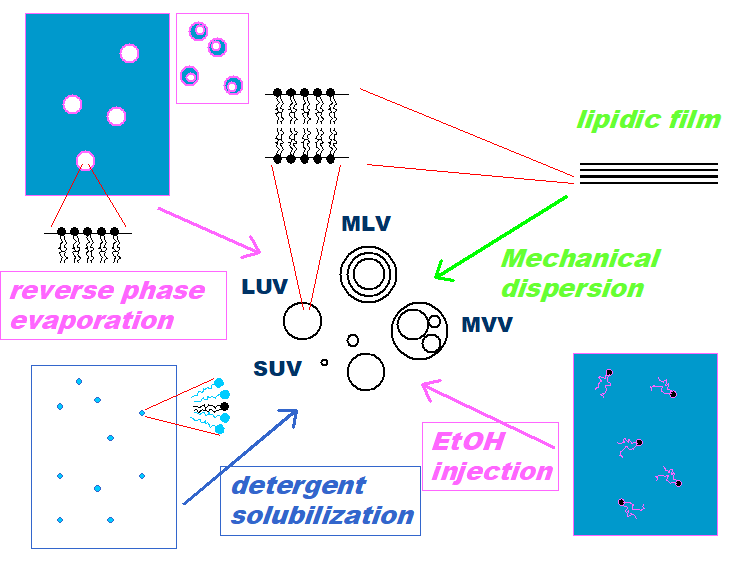
Many methods
Lipid-film hydration
To ensure complete and homogenous mixing of all the lipidic components as they are required in the final lipid vesicles preparation we dissolve the lipids mixture together in the same solvent solution. After obtained a homogenous solution of these lipids we dry down them in a rotary evaporator. The temperature for drying down should be regulated so that it is above the phase transition temperature of the lipid mix. A thin lipid film is formed.

Extrusion technique
This is a gentle method of reducing the size of lipid vesicles. It consists to pass them through a membrane filter of defined pore size at low pressure by using LiposoFast device, giving populations in which one can choose the upper size limit depending on the exact pore size of the filter used. VET vesicles with dimension from 100 to 400 nm can be obtained.


Detergent dialysis method
In principle the phospholipid is brought into intimate contact with the aqueous phase via the intermediary of detergents, which associate with phospholipids molecules and serve to screen the hydrophobic portions of the molecule from water providing their concentration is high enough, this is the critical micellar concentration (CMC), below this concentration the detergent molecules exist entirely in free solution. A way to remove the detergent molecules is by controlled detergent removal through dialysis. The molecules of detergent can be removed from the mixed micelles upon lowering the concentration of detergent in the bulk aqueous phase by dialysis throughout a membrane with cut off around 10000 dalton.



